ファーマコビジランスあるいは医薬品安全性管理とは「医薬品の有害作用やその他の医薬品関連の問題を検出し、評価し、理解し、そして、予防するための科学・活動」と定義されています。安全で適切な医薬品を患者に提供するため、ファーマコビジランスは重要な役割を果たしています(Lakshmi, Aashritha, Teja, & Sridhar, 2016)。製薬会社や規制当局は、患者の安全確保のため、ファーマコビジランスのツールとしてシグナル検出(信号検出)の技術を利用して医薬品のリスク増大に関わるデータを積極的に分析しています。
ファーマコビジランスのシグナル検出は潜在的に存在する医薬品の有害反応の情報を信号として特定・調査する作業です。この場合、シグナルを特定する手段としてはファーマコビジランスのデータベース、医学文献、ソーシャルメディア、医療専門家や患者などからの自発的な報告など、さまざまな情報源を利用できます。
ただし、ファーマコビジランスデータはセンシティブな情報を含み、さらには、規制要件とも関わるため、アクセスが制限されたり、情報の信頼性に問題がある場合もあります(Kumar & Khan、2015)。ファーマコビジランスは進化の途中であり、データの収集・処理、予測、データセキュリティおよびシグナル検出についてAI化が進んでいます。
「人工知能(AI)とは人間が行う知的作業をデジタルコンピュータが担うことである。」(Copeland、2023)。Copelandは、過去の経験に基づいて推論する、意味を見つける、一般化する、学習するなどの人間の能力を機械に備えることは可能であると言っています。機械学習とは、明示的なプログラミングによらない方法で新しい学習能力を機械に組み込むこと(Das S、2015)、また、あるタスクを行うアルゴリズムを成功と失敗を繰り返して学習しながら獲得すること(Hashimoto DA、2018)です。医療システムのAI化は医療提供者の役割を変え、そして、患者の安全性の向上(Macrae C、2019)と医療の質の向上をもたらす可能性があります。ファーマコビジランスの分野では、安全性の確保、対象集団の特定、シグナル管理などのさまざまな場面でAIが広く受け入れられ、求められています。

AIは急速に成長している技術分野であり、ファーマコビジランスへの応用面では大きな可能性を秘めています。現在、個別症例安全性報告の件数が年々増加していて、有害事象の90%が報告されていない状況です。患者の安全を確保するためには技術の改善が必要です。AIと人間を比較した場合、データの無欠性の確保、データマイニング、法令遵守などの面で、AIはデータの品質を向上させることができる、処理中にリアルタイムにデータを考察することができる、迅速かつプロアクティブにシグナル検出を行うことができるなど、人間の専門家よりも優れています(Traverso、2020)。AIは複雑な状況下での意思決定をサポートしてくれます(Salas1 M、2022)。例えば、AIにデータマイニングを行わせた場合、大量のデータを収集・評価し、そして、リスク評価のプロセスを支援するため、患者の安全性が向上します。
ファーマコビジランスへのAIの応用分野は複数あります。例えばADE(薬物有害事象)やADR(薬物有害反応)の特定、医薬品市販後サーベイランス、安全性報告書内の自由記述の分類(Bates DW、2021)、薬物間相互作用の検出、薬物毒性ハイリスク集団の特定、薬物副作用の予測、臨床試験のシミュレーション、シグナル検出などです。
シグナル検出は医薬品使用および安全性サーベイランスの不可欠な要素です(R H Meyboom 1、1997)。個別症例安全性報告(ICSR)用のシグナル検出は「定性的」に行うこともできますが、現実世界のデータベースと機械学習、またはデータマイニングの手法を適用することによって「定量的」に行うことが可能です。定量的または定性的に検出したシグナルは臨床判断による検証と確認が必要です。シグナルは新たな安全性情報を示すものと、既知の薬物有害反応の新たな側面を示すもののいずれの場合もありますが、いずれにしても薬剤との因果関係を示すものです。そして、その因果関係の真偽を確かめるためのさらなる調査が必要となります。これらの医薬品の安全性の問題はICSR、自発報告システム、科学文献、生物医学的データベース、医療記録などの様々なデータソースから見つけることができます。
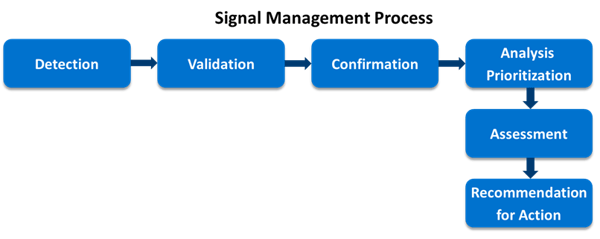
医薬品開発プロセスにおいて、シグナル検出は患者の安全を確保する上で不可欠な要素です。問題を顕在化する前に早期に発見できれば、患者の安全性を向上できるばかりでなく、高騰を続ける医薬品の経済的負担を軽減し、さらに、好ましくない有害事象も減らすことができます。シグナルは患者の苦痛のエビデンスであり、それに基づいて研究を行えば、将来、患者の苦痛を軽減できるかもしれません。しかし、ファーマコビジランスによっても未知の予期せぬシグナルを市販後早期に検出することは容易ではありません(Ritu Rani、2019)。
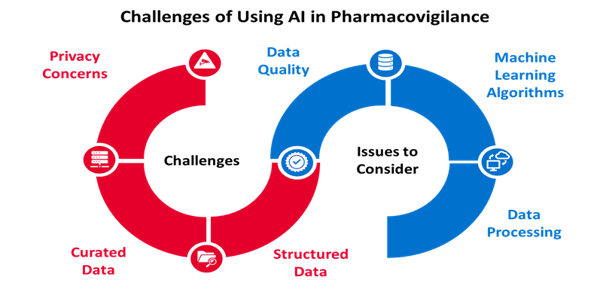
ファーマコビジランスは医療に不可欠な重要な機能です。しかし、この分野での人工知能の利用は比較的新しく、発展途上です。医薬品の安全問題を特定する手段としてAIを使用するにあたっては、ソフトウェアのトレーニング用として適切に構築され整理されたデータベースを使えるかどうかが大きな課題です。ファーマコビジランスのAI化にあたっては、個人情報が本人の同意なしに他の目的に使用される可能性があることから、プライバシーの問題が懸念されます。ファーマコビジランスのシステムは情報の欠落があった場合にフラグメンテーションを起す可能性があり、その場合は機械学習が困難になったり、一貫性を保てなくなる恐れがあります。そのため、多くの企業は自社のビジネスモデルにAIを導入するにあたって、段階的に導入するアプローチをとっています。ファーマコビジランスにAIを導入する場合、最初に安全性データベースにAIを使う方法があります。この場合、AIは、データの一貫性を確保しながら、データ収集と症例比較の作業をサポートしてくれます。ファーマコビジランスチームはAIから提供されるデータとデータソースを比較することによってマシンの品質をチェックすることができます。AIを一旦導入した後は、文章の自動生成、症例処理、シグナル検出のための安全データの分析など、さらに複雑なプロセスにAIを導入することができます。人工知能は大量のデータを管理し、そして、完全かつ正確に分析することができますから、その意味でファーマコビジランスの将来を担っていると言えるでしょう。
※本記事は2023年10月19日に発信した英文記事の翻訳をもとに作成したものです。
文責:
Bawneet Narang
Pharmacovigilance Associate – Linical
参考文献:
- Bates, D.W., Levine, D., Syrowatka, A., Kuznetsova, M., Craig, K.J.T., Rui, A., et al (2021). The potential of artificial intelligence to improve patient safety: a scoping review. NPJ Digital Medicine, 4, 54.
- Macrae, C. (2019). Governing the safety of artificial intelligence in healthcare. BMJ Quality Safety, 28(6), 495–498.
- Copeland, B. (2023, APR 25). Artificial Intelligence. Retrieved APR 25, 2023, from Britannica: https://www.britannica.com/technology/artificial-intelligence.
- Das, S., Dey, A., Pal, A., Roy, N. (2015). Applications of artificial intelligence in machine learning: review and prospect. International Journal of Computer Applications, 115, 31-41.
- Hashimoto, D.A., Rosman, G., Rus, D., Meireles, O.R (2018). Artificial intelligence in surgery: promises and perils. Annals of Surgery, 268(1), 70–76.
- Kumar, A., Khan, H. (2015). Signal Detection and their Assessment in Pharmacovigilance. Open Pharmaceutical Sciences Journal, 2, 66-73.
- Lakshmi, A., Aashritha, M., Teja, K., Sridhar, R. (2016). A Review On Pharmacovigilance and Its Importance. World Journal of pharmacy and pharmaceutical sciences, 6(1), 300-310.
- Meyboom, R.H., Egberts, A.C., Edwards, I.R., Hekster, Y.A., Koning, F.H. de., Gribnau, F.W. (1997). Principles of signal detection in pharmacovigilance. National Library of Medicine, 16(6), 355-365.
- Rani, R., Chand, S., Devi, M., Singh, A., Kumar, D. (2019). A Review on Signal in Pharmacovigilance. Asian Journal of Pharmaceutical Research and Development, 7(6), 80-84.
- Salas, M., Petracek, J., Yalamanchili, P., Aimer, O., Kasthuril, D., Dhingra, S., et al (2022). The Use of Artifcial Intelligence in Pharmacovigilance: A Systematic Review of the Literature. Pharmaceutical Medicine, 36 (5), 295-306.
- Traverso, K., Markey, J. (Aug 2020). The Untapped Potential of AI & Automation in Pharmacovigilance. Pharmaceutical Engineering. Retrieved APR 26, 2023: https://ispe.org/pharmaceutical-engineering/july-august-2020/untapped-potential-ai-automation-pharmacovigilance
.jpg)